Electroplating vs electroforming: what's the difference?
Jul 4, 2017 8:00:00 AM By Harrie KnolElectroplating and electroforming are not the same. Sure, the title of this article gave that away already, but it is a common misunderstanding. That is why we take the time to explain the differences and similarities between both processes. Be careful though: whoever discovers electroforming and electroplating enters a whole new world of ultra-precision metal applications.
To learn more about the technology and discover the potential of electroforming: download our electroforming whitepaper here
The electrodeposition process: what is it, and how does it work?
First things first. Both electroplating and electroforming are conducted through an electrodeposition process. In short, this means deposition of metal onto a conductive object. Both an anode and a cathode are immersed in an electrolytic bath that is composed of a solution (ion) of salt and the metal to be plated. A direct current (DC) of electricity is passed through the solution, effecting the transfer of metal ions onto a cathodic surface, plating the metal onto the item. Keeping this in mind, we will now zoom in on electroforming and electroplating.
The electroforming process explained
Electroforming is the metal forming process where metal is grown by electrodeposition onto a substrate. The electrolytic bath is used to deposit metal (for example nickel, gold or copper) onto a conductive patterned surface.
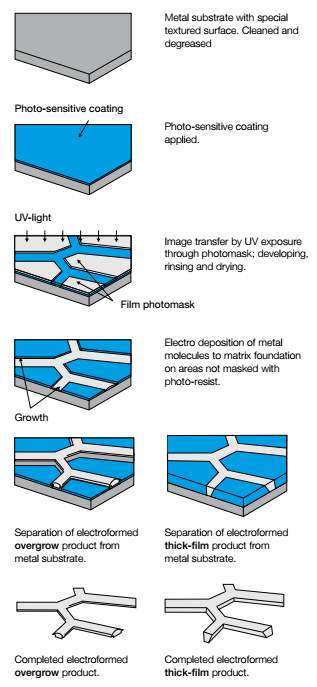
Fig.1: The electroforming process
In electroforming, the metal can be grown as thick as the customer wants. In the industry, the model on which the metal is built is called a mandrel. The mandrel is typically removed after the completion of the plating process. The mandrel is made from conductive and nonconductive area’s. The metal will only deposit on the conductive area’s. The metal can easily be removed from the mandrel. The result is an autonomous product with holes, slots or any shape you prefer. Read more in our latest electroforming whitepaper.

The electroplating process explained
In electroplating, a very thin layer is deposited on an existent object by means of the electrodeposition process. It can also be applied after the electroforming process. Electroplating can be applied on a broad range of products. The materials used in the plating (coating) process depend on the composition of the plating bath and the deposition conditions. Electroplating can be used to change the surface property of the object. For example, to create a better resistance against chemical and/or abrasive corrosion.
Electroplating vs electroforming: what's the difference?
So if both processes are done by means of electrodeposition, what is the main difference between the two? In electroplating, the deposited metal becomes part of the existing product by plating/coating it. In electroforming, the mandrel (patterned surface) will be removed from the product. After the mandrel is removed, the object that remains is entirely created through electrodeposition. After electroforming it is possible to perform electroplating to add a coating to improve corrosion-resistance or to get a more attractive (cosmetic) product.
Electroforming: why you should consider it as an engineer
There are a lot of benefits that come with electroforming, and some of them particularly solve the challenges you have as an engineer. Short lead times, high operational efficiency, continuous innovation, cost-effective and large-scale production — you can accomplish these goals by utilizing the right manufacturing method. Electroforming may just be the technique that will help you drive true innovation.
If you’d like to learn more about electroforming — and want to find out if it’s the technique that fits your engineering needs — read our latest electroforming whitepaper:
